Lucid Dreaming: Psychophysiological Studies
Lucid Dreaming: Psychophysiological Studies of Consciousness during REM Sleep
by Stephen LaBerge, Ph.D.
From: http://www.lucidity.com/SleepAndCognition.html
Lucid Dreaming Physiologically Verified
Although we are not usually explicitly aware of the fact that we are dreaming while we are dreaming, at times a remarkable exception occurs, and we become conscious enough to realize that we are dreaming. "Lucid" dreamers (the term derives from van Eeden, 1913) report being able to freely remember the circumstances of waking life, to think clearly, and to act deliberately upon reflection, all while experiencing a dream world that seems vividly real (Green, 1968; LaBerge, 1985; Gackenbach & LaBerge, 1988). This is all in contrast to the usual past characterization of dreams as typically lacking any reflective awareness or true volition (Rechtschaffen, 1978).
Lucid dreaming is normally a rare experience. Though most people report having had a lucid dream at least once in their lives, only about 20% of the population reports having lucid dreams once a month or more (Snyder & Gackenbach, 1988).
In spite of the fact that most people have experienced lucid dreams, some theoreticians have considered them impossible and even absurd (eg, Malcolm, 1959). In the absence of empirical evidence bearing on the question, most sleep researchers were apparently inclined to accept Hartmann's "impression" that lucid dreams were "not typical parts of dreaming thought, but rather brief arousals" (Hartmann, 1975, p. 74; Berger, 1977). Schwartz and Lefebvre (1973) noted that frequent transitory arousals were common during REM sleep and proposed these "micro-awakenings" as the physiological basis for lucid dream reports. Although no one had put forward any evidence for this mechanism, it seems to have been the predominant opinion (cf. Foulkes, 1974) up until the last few years.
Empirical evidence began to appear in the late 1970s suggesting that lucid dreams occur during REM sleep. Based on standard sleep recordings of two subjects who reported a total of three lucid dreams upon awakening from REM periods, Ogilvie, Hunt, Sawicki, and McGowan (1978) cautiously concluded that "...it may be that lucid dreams begin in REM." However, no proof was given that the reported lucid dreams themselves had in fact occurred during the REM sleep immediately preceding the awakenings and reports. What was needed to unambiguously establish the physiological status of lucid dreams was some sort of behavioral response signaling to the experimenter the exact time the lucid dream was taking place.
We provided the necessary verification by instructing subjects to signal the onset of lucid dreams with specific dream actions that would be observable on a polygraph (i.e., eye movements and fist clenches). Using this approach, LaBerge, Nagel, Dement & Zarcone (1981) reported that the occurrence of lucid dreaming during unequivocal REM sleep had been demonstrated for five subjects. After being instructed in the method of lucid dream induction (MILD) described by LaBerge (1980b) the subjects were recorded from 2 to 20 nights each. In the course of the 34 nights of the study, 35 lucid dreams were reported subsequent to spontaneous awakening from various stages of sleep as follows: REM sleep 32 times, NREM Stage-1, twice, and during the transition from NREM Stage-2 to REM, once. The subjects reported signaling during 30 of these lucid dreams. After each recording, the reports mentioning signals were submitted along with the respective polysomnograms to a judge uninformed of the times of the reports. In 24 cases (90%), the judge was able to select the appropriate 30 second epoch on the basis of correspondence between reported and observed signals. All signals associated with lucid dream reports occurred during epochs of unambiguous REM sleep scored according to the conventional criteria (Rechtschaffen & Kales, 1968).
A later analysis extending these data with two additional subjects and 20 more lucid dreams produced identical results (LaBerge, Nagel, Taylor, Dement, & Zarcone, 1981). LaBerge et al. argued that their investigations demonstrated that lucid dreaming usually (though perhaps not exclusively) occurs during REM sleep. This conclusion is supported by research carried out in several other laboratories (Dane, 1984; Fenwick et al., 1984; Hearne, 1978; Ogilvie, Hunt, Kushniruk, & Newman, 1983).
Ogilvie et al. (1983) reported the physiological state preceding 14 spontaneous lucidity signals as unqualified REM in 12 (86%) of the cases; of the remaining two cases, one was "ambiguous" REM and the other appeared to be wakefulness. Keith Hearne and Alan Worsley collaborated on a pioneering study of lucid dreaming in which the latter spent 50 nonconsecutive nights in the Hull University sleep lab while the former monitored the polygraph. Worsley reported signaling in 8 lucid dreams, all of which were described by Hearne (1978) as having occurred during REM sleep.
However, demonstrations that signaling of lucid dreams occurs during REM sleep raises another kind of question: What exactly do we mean by the assertion that lucid dreamers are 'asleep?' Perhaps these 'dreamers' are not really dreamers, as some argued in the last century; or perhaps this 'sleep' is not really sleep, as some have argued in this century. How do we know that lucid dreamers are 'really asleep' when they signal? If we consider perception of the external world as a criterion of being awake (to the external world), we can conclude that they are actually asleep (to the external world) because although they know they are in the laboratory, this knowledge is a matter of memory, not perception. Upon awakening, they report having been totally in the dream world and not in sensory contact with the external world.
It might be objected that lucid dreamers might simply not be attending to the environment; rather than being asleep, perhaps they are merely absorbed in their private fantasy worlds as, for example, when deeply immersed in a novel or daydream. However, according to the reports of lucid dreamers (LaBerge, 1980a, 1985), if they deliberately attempt to feel the bedcovers they know they are sleeping in or try to hear the ticking of the clock they know is beside their bed, they fail to feel or hear anything except what they find in their dream worlds. Lucid dreamers are conscious of the absence of sensory input from the external world; therefore, on empirical grounds, they conclude that they are asleep.
If, in a contrary case, subjects were to claim to have been awake while showing physiological signs of sleep, or vice versa, we might have cause to doubt their subjective reports. However, when -- as in the present case -- the subjective accounts and objective physiological measures are in clear agreement, it is embarrassingly awkward to assert (as some critics have done) that subjects who reported being certain that they were asleep while showing physiological indications of unequivocal sleep were actually awake (cf. LaBerge, Nagel, Dement & Zarcone, 1981).
Some critics have suggested that "demand characteristics" might account for our results. It is true that our subjects were under demand to have, signal and report lucid dreams, but how could demand alone account for them doing all three things without having been lucid in the first place? If they merely unconsiously signaled, we would have found REM periods with signals without subsequent reports of lucidity -- but we did not. If they merely reported having signaled without actually having done so, we would have found reports without signals, which we did not. Further, by this account, where would the reported and observed signals have come from?
The evidence is clear: lucid dreaming is an experiential and physiological reality; though perhaps paradoxical, it is clearly a phenomenon of sleep.
Physiological Characteristics Of Lucid Dreaming
The preceding studies have shown that lucid dreams typically occur in REM sleep. However, since REM sleep is a heterogeneous state exhibiting considerable variations in physiological activity, of which two distinct phases are ordinarily distinguished. In its most active form, REM is dominated by a striking variety of irregular and short-lived events such as muscular twitching, including the rapid eye movements that give the state one of its most common names. This variety of REM is referred to as 'phasic,' while the relatively quiescent state remaining when rapid eye movements and other phasic events temporarily subside is referred to as 'tonic.' On first thought, one might expect lucid dreams to be associated with decreased phasic activity (Pivik, 1986). However, research by the Stanford group, detailed below, has shown lucid dreaming to be associated with, on the contrary, increased phasic activity.
LaBerge, Levitan, and Dement (1986) analyzed physiological data from 76 signal-verified lucid dreams (SVLDs) of 13 subjects. The polysomnograms corresponding to each of the SVLDs were scored for sleep stages and every SVLD REM period was divided into 30 s epochs aligned with the lucidity onset signal. For each epoch, sleep stage was scored and rapid eye movements (EM) were counted; if scalp skin-potential responses were observable as artifacts in the EEG, these were also counted (SP). Heart rate (HR) and respiration rate (RR) were determined for SVLDs recorded with these measures.
For the first lucid epoch, beginning with the initiation of the signal, the sleep stage was unequivocal REM in 70 cases (92%). The remaining six SVLDs were less than 30 s long and hence technically unscorable "by the book" (Rechtschaffen & Kales, 1968). For these cases, the entire SVLD was scored as a single epoch; with this modification, all SVLDs qualified as REM. The lucid dream signals were followed by an average of 115 s (range: 5 to 490 s) of uninterrupted REM sleep. Physiological comparison of EM, HR, RR, and SP for lucid vs. non-lucid epochs revealed that the lucid epochs of the SVLD REM periods had significantly higher levels of physiological activation than the preceding epochs of non-lucid REM from the same REM period. Similarly, H-reflex amplitude is lower during lucid compared to non-lucid REM (Brylowski, Levitan, & LaBerge, 1989).
In order to study the temporal variations of physiology as they correlated with the development and initiation of lucidity, for each SVLD REM period the physiological variables were converted to standard scores and averaged across dreams and subjects. Figure 1 is a histogram of the resultant mean standard scores for the five minutes before and the five minutes after the initiation of lucidity. Note the highly significant increases in physiological activation during the 30 s before and after lucidity onset.
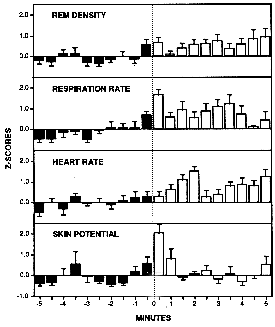
Figure 1. Histograms of grand mean z-scores for EM, RR, HR, and SP.Bins are 30 s in length with t=0 representing the signaled onset of lucidity. Ns vary with variable and bin, but all values are averaged across lucid dreams and subjects. (* p<.05)
Physiological data (EM, RR, HR, and SP) were also collected for sixty-one control non-lucid REM periods, derived from the same 13 subjects, in order to allow comparison with SVLDs. Mean values for EM and SP were significantly higher for REM periods with lucid dreams than non-lucid control REM periods (RR and HR did not differ).
Given the finding that lucid dreams reliably occur during activated (phasic) REM, measures of central nervous system activation, such as eye movement density, should contribute something to the pattern of lucid dream distribution. Since it had been previously observed that eye-movement density starts at a low level at the beginning of REM periods and increases until it reaches a peak after approximately five to seven minutes (Aserinsky, 1971), we (LaBerge et al., 1986) hypothesized that lucid dream probability should follow a parallel development and accordingly found that mean eye-movement density correlated positively and significantly with lucid dream probability (r = .66, p < .01).
Lucid dreams have been frequently reported to occur most commonly late in the sleep cycle (Green, 1968). LaBerge et al. (1986) tested this hypothesis by first determining for each of their 12 subjects the time of night which divided their total REM time into two equal parts. All but one of the subjects had more lucid dreams in the second half of their REM time than in the first half (binomial test; p < .01). For the combined sample, relative lucidity probability was calculated for REM periods one through six of the night by dividing the total number of lucid dreams observed in a given REM period by the corresponding total time in stage REM for the same REM period. A regression analysis clearly demonstrated that relative lucidity probability was a linear function of ordinal REM period number (r = .98, p < .0001).
There are two distinct ways in which lucid dreams are initiated. In the usual case, subjects report having been in the midst of a dream when a bizarre occurrence causes sufficient reflection to yield the realization that they are dreaming. In the other, less frequent case, subjects report having briefly awakened from a dream and then falling back asleep directly entering the dream with no (or very little) break in consciousness (Green, 1968; LaBerge 1985a). Here is an example of a wake-initiated lucid dream:
I was lying awake in bed late in the morning listening to the sound of running water in the adjoining bathroom. Presently an image of the ocean appeared, dim at first like my usual waking imagery. But its vividness rapidly increased while, at the same time, the sound of running water diminished; the intensity of the internal image and external sound seemed to alter inversely (as if one changed a stereo balance control from one channel to the other). In a few seconds, I found myself at the seashore standing between my mother and a girl who seemed somehow familiar. I could no longer hear the sound of the bath water, but only the roar of the dream sea.... (LaBerge, 1980, p. 85)
Note that the subject is continuously conscious during the transition from wakefulness to sleep. This fact suggests that Foulkes (1985) is over-stating the case by claiming that it is "...a necessary part of the experience we call 'sleep' that we lose a directive and reflective self. You can't fall asleep, or be asleep, if your waking self is still regulating and reflecting upon your conscious mental state" (p. 42).
Since lucid dreams initiated in these two ways ought to differ physiologically in at least one respect (i.e., an awakening preceding one but not the other), the SVLDs were dichotomously classified as either 'Wake-initiated' (WILD) or 'Dream-initiated' (DILD), depending on whether or not the reports mentioned a transient awakening in which the subject consciously perceived the external environment before re-entering the dream state.
Fifty-five (72%) of the SVLDs were classified as DILDs and the remaining 21 (28%) as WILDs. For all 13 subjects, DILDs were more common than WILDs (binomial test, p < .0001). As expected, compared to DILDs, WILDs were more frequently immediately preceded by physiological indications of awakening (Chi-squared = 38.3, 1df, p < .0001) establishing the validity of classifying lucid dreams in this manner. See Figures 2 and 3 for illustrations of these two types of lucid dream.
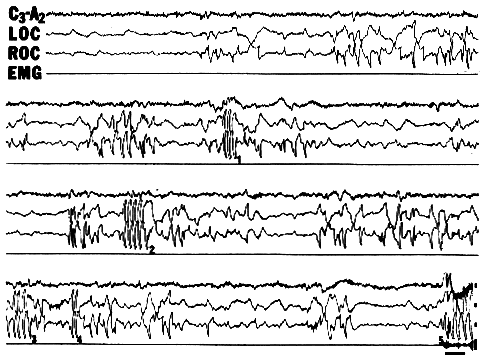
Figure 2. A typical dream-initiated lucid dream (DILD). Four channels of physiological data (central EEG [C3-A2], left and right eye-movements [LOC and ROC], and chin muscle tone [EMG]) from the last 8 min of a 30 min REM period are shown. Upon awakening the subject reported having made five eye movement signals (labeled 1-5 in figure). The first signal (1, LRLR) marked the onset of lucidity. Skin potential potential artifacts can be observed in the EEG at this point. During the following 90 s the subject "flew about" exploring his dream world until he believed he had awakened, at which point he made the signal for awakening (2, LRLRLRLR). After another 90 s, the subject realized he was still dreaming and signaled (3) with three pairs of eye movements. Realizing that this was too many, he correctly signaled with two pairs (4). Finally, upon awakening 100 s later he signaled appropriately (5, LRLRLRLR). [Calibrations are 50 microV and 5 s.]
As was mentioned earlier, momentary intrusions of wakefulness occur very commonly during the normal course of REM sleep and it had been proposed by Schwartz and Lefebvre (1973) that lucid dreaming occurs during these micro-awakenings. However, LaBerge et al.'s (1981,1986) data indicates that while lucid dreams do not take place during interludes of wakefulness within REM periods, a minority of lucid dreams (WILDs) are initiated from these moments of transitory arousal, with the WILDs continuing in subsequent undisturbed REM sleep.
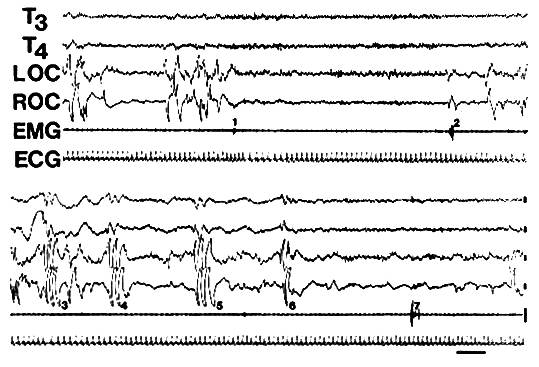
Figure 3. A typical lucid dream initiated from a transient awakening during REM (WILD). Six channels of physiological data (left and right temporal EEG [T3 and T4], left and right eye-movements [LOC and ROC], chin muscle tone [EMG], and electrocardiogram [ECG]) from the last 3 min of a 14 min REM period are shown. The subject awoke at 1 and after 40 s returned to REM sleep at 2, and realized he was dreaming 15 s later and signaled at 3. Next he carried out the agreed-upon experimental task in his lucid dream, singing between signals 3 and 4, and counting between signals 4 and 5. This allowed comparison of left and right hemisphere activation during the two tasks (LaBerge and Dement, 1982b). Note the heart-rate acceleration-deceleration pattern at awakening (1) and at lucidity onset (3), and the skin potential potential artifacts in the EEG (particularly T4) at lucidity onset (3). [Calibrations are 50 microV and 5 s.]
To summarize, an elevated level of CNS activation seems to be a necessary condition for the occurrence of lucid dreams. Evidently the high level of cognitive function involved in lucid dreaming requires a correspondingly high level of neuronal activation. In terms of Antrobus's (1986) adaptation of Anderson's (1983) ACT* model of cognition to dreaming, working memory capacity is proportional to cognitive activation, which in turn is proportional to cortical activation. Becoming lucid requires an adequate level of working memory to activate the pre-sleep intention to recognize that one is dreaming. This level of cortical and cognitive activation is apparently not always available during sleep, but normally only during phasic REM.
Psychophysiological Relationships During REM Sleep
Psychologists attempting to apply rigorous scientific methodology to the study of such phenomena as mental imagery, hallucinations, dreaming, and in general, conscious processes face a major challenge: The most direct account available of the private events occurring in a person's mind is his or her own subjective report. But, unfortunately, subjective reports are difficult to objectively verify and introspection is far from an unbiased and direct process of observation. There are two strategies likely to increase our confidence in the reliability of subjective reports: 1) use highly trained (and in the context of dream research, lucid) subjects who are skillful reporters. 2) use the psychophysiological approach, which makes use of the fact that the convergent agreement of physiological measures and subjective reports provides a degree of validation to the latter (Stoyva and Kamiya, 1968).
Indeed, the psychophysiological approach was responsible for the Golden Age of dream research in the decades following the discovery of REM sleep (Aserinsky and Kleitman, 1953) and the subsequent association of REM with dreaming (Dement and Kleitman, 1957). Although the psychophysiological paradigm of dream research yielded an abundant harvest for many years (see Arkin, Antrobus & Ellman, 1978), it possessed a fatal flaw: as long as the subjects are non-lucid, the researcher has no way of making certain that the subjects will dream about what the researcher might like to study. Pre-sleep manipulations producing reliable effects on dream content have not been highly successful (Tart, 1988). One can only wait and hope that eventually a dream report will turn up what one is looking for. This is really no better than a shot-in-the-dark approach and some researchers have been calling for abandoning the psychophysiological method in favor of a purely psychological approach. An influential researcher has written that "...psychophysiological correlation research now appears to offer such a low rate of return for effort expended as not to be a wise place for dream psychology to continue to commit much of its limited resources" (Foulkes, 1981, p. 249). This conclusion may well be justified, but only insofar as it refers to the psychophysiological approach as traditionally practiced, using non-lucid subjects. The use of lucid dreamers overcomes the basic difficulty of the old methodology, and may revitalize the psychophysiological approach to dream research.
The fact that lucid dreamers can remember to perform predetermined actions and signal to the laboratory suggested to LaBerge (1980a) a new paradigm for dream research: Lucid dreamers, he proposed, "could carry out diverse dream experiments marking the exact time of particular dream events, allowing the derivation of precise psychophysiological correlations and the methodical testing of hypotheses" (LaBerge, Nagel, Dement, & Zarcone, 1981, p. 727). This strategy has been put into practice by the Stanford group in a number of studies summarized below.
How long do dreams take? This question has intrigued humanity for many centuries. A traditional answer was that dreams take very little or no time at all, as in the case of Maury's famous dream in which he had somehow gotten mixed up in a long series of adventures during the French Revolution, finally losing his head on the guillotine, at which point he awoke to find the headboard had fallen on his neck. He supposed, therefore that the lengthy dream had been produced in a flash by the painful stimulus. The idea that dreams occur in the moment of awakening has found supporters over the years (e.g., Hall,1981).
We have straightforwardly approached the problem of dream time by asking subjects to estimate ten second intervals (by counting, "one thousand and one, one thousand and two, etc.") during their lucid dreams. Signals marking the beginning and end of the subjective intervals allowed comparison with objective time. In all cases, time estimates during the lucid dreams were very close to the actual time between signals (LaBerge, 1980a, 1985). However, this finding does not rule out the possibility of time distortion effects under some circumstances.
The data reported by LaBerge, Nagel, Dement, and Zarcone (1981) and LaBerge, Nagel, Taylor, Dement, and Zarcone (1981) indicate that there is a very direct and reliable relationship between gaze shift reported in lucid dreams and the direction of polygraphically recorded eye movements. The results obtained for lucid dreams (see also Dane, 1984; Fenwick et al., 1984; Hearne, 1978; Ogilvie, Hunt, Tyson, Lucescu, & Jeakins, 1982) are much stronger than the generally weak correlations obtained by previous investigators testing the hypothesis that the dreamer's eyes move with his or her hallucinated dream gaze, who had to rely on the chance occurrence of a highly recognizable eye-movement pattern that was readily matchable to the subject's reported dream activity (e.g. Roffwarg, Dement, Muzio, & Fisher, 1962).
LaBerge (1986) has carried out related experiments in which two subjects tracked the tip of their fingers moving slowly left to right during four conditions: 1) awake, eyes open; 2) awake, eyes closed mental imagery; 3) lucid dreaming; and 4) imagination ("dream eyes closed") during lucid dreaming. The subjects showed saccadic eye movements in the two imagination conditions (2 and 4), and smooth tracking eye movements during dreamed or actual tracking (conditions 1 and 3).
In another study, LaBerge and Dement (1982a) demonstrated the possibility of voluntary control of respiration during lucid dreaming. They recorded three lucid dreamers who were asked to either breathe rapidly or to hold their breath (in their lucid dreams), marking the interval of altered respiration with eye movement signals. The subjects reported successfully carrying out the agreed-upon tasks a total of nine times, and in every case, a judge was able to correctly predict on the basis of the polygraph recordings which of the two patterns had been executed (binomial test, p < .002).
Evidence of voluntary control of other muscle groups during REM was found by LaBerge, Nagel, Dement, and Zarcone (1981) while testing a variety of lucidity signals. They observed that a sequence of left and right dream-fist clenches resulted in a corresponding sequence of left and right forearm twitches as measured by EMG. However, the amplitude of the twitches bore an unreliable relationship to the subjective intensity of the dreamed action. Because all skeletal muscle groups except those that govern eye-movements and breathing are profoundly inhibited during REM sleep for, it is to be expected that most muscular responses to dreamed movements will be feeble. Nonetheless, these responses faithfully reflect the motor patterns of the original dream. Similar observations have been made by Fenwick et al. (1984).
Following reports of cognitive task dependency of lateralization of EEG alpha activity in the waking state by many researchers, LaBerge & Dement (1982b) undertook a pilot study demonstrating the feasibility of similar investigations in the lucid dream state. The two tasks selected for comparison were dreamed singing and dreamed counting, activities expected to result in relatively greater engagement of the subjects' left and right cerebral hemispheres, respectively.
Integrated alpha band EEG activity was derived from electrodes placed over right and left temporal lobes while four subjects sang and estimated 10 seconds by counting in their lucid dreams (marking the beginning and end of each task by eye movement signals). The results supported the hypothesized lateralization of alpha activity: the right hemisphere was more active than the left during singing; during counting the reverse was true. These shifts were similar to those observed during actual singing and counting. In contrast, a control condition with imagined singing and counting showed no significant laterality shifts. Because of the small number of subjects, the conclusions of this study must be regarded as suggestive at best.
LaBerge & Dement noted an important implication of their results for the interpretation of EEG alpha activity during REM sleep. Since continuous alpha activity occurs when a subject awakens, sleep researchers have usually assumed that increased alpha activity in the context of sleep is always a sign of wakefulness or relative cortical activation. The findings just discussed suggest the contrary: alpha activity during REM sleep is, as in waking, inversely related to cortical activation. When a person awakens from a vivid dream to a dark room, his cortical (occipital, at least) activation has decreased, not increased, with the resultant appearance of elevated alpha power.
In this view, it is a straightforward prediction that occipital alpha power during REM sleep will correlate negatively with subsequently reported dream vividness. This could provide the proper explanation for the finding that awakenings following REM periods with high levels of alpha activity are more likely to yield "thinking" reports than awakenings from low-alpha REM periods which yield more "dreaming" reports (Antrobus, Dement & Fisher (1964).
Sexual activity is a rather commonly reported theme of lucid dreams (Garfield, 1979; LaBerge, 1985). LaBerge, Greenleaf, and Kedzierski (1983) undertook a pilot study to determine the extent to which subjectively experienced sexual activity during REM lucid dreaming would be reflected in physiological responses.
Sixteen channels of physiological data, including EEG, EOG, EMG, respiration, skin conductance level (SCL), heart rate, vaginal EMG (VEMG) and vaginal pulse amplitude (VPA), were recorded from a single subject. The experimental protocol called for her to make specific eye movement signals at the following points: when she realized she was dreaming (i.e., the onset of the lucid dream); when she began sexual activity (in the dream); and when she experienced orgasm.
The subject reported a lucid dream in which she carried out the experimental task exactly as agreed upon. Data analysis revealed a significant correspondence between her subjective report and all but one of the autonomic measures; during the 15 second orgasm epoch, mean levels for VEMG activity, VPA, SCL, and respiration rate reached their highest values and were significantly elevated compared to means for other REM epochs. Contrary to expectation, heart rate increased only slightly and non-significantly.
Implications for Research on Sleep and Cognition
The fact of lucid dreaming presents conceptual difficulties for certain traditional beliefs about "sleep" and presumed limitations of dream mentation. In a certain sense, the anomalous appearance of lucid dreaming parallels that of the state that has been called "paradoxical sleep." The discovery of REM sleep required the expansion of our concept of sleep. The evidence associating lucid dreaming with REM sleep reviewed above would seem to require a similar expansion of our concept of dreaming, and a clarification of our concept of sleep.
Fenwick et al. (1984) showed that a subject was able to perceive and respond to environmental stimuli (electrical shocks) without awakening from his lucid dream. This result raises a theoretical issue: if we take perception of the external world to be the essential criterion for wakefulness (LaBerge et al., 1981a; see above) then it would seem that Worsley must have been at least partially awake. On the other hand, when environmental stimuli are incorporated into dreams without producing any subjective or physiological indications of arousal, it appears reasonable to speak of the perception as having occurred during sleep.
Furthermore, it may be possible, as LaBerge (1980c) has suggested, for one sense to remain functional and 'awake' while others fall 'asleep.' Similarly, Antrobus, Antrobus and Fisher (1965) argued "...that the question -- awake or asleep -- is not a particularly useful one. Even though we have two discrete words -- sleep and wakefulness -- this does not mean that the behavior associated with the words can be forced into two discrete categories. ... not only do sleeping and waking shade gradually into one another but there is only limited agreement among the various physiological and subjective operations that discriminate between sleeping and waking. At any given moment, all systems of the organism are not necessarily equally asleep or awake." (pp. 398-399)
As long as we continue to consider wakefulness and sleep as a simple dichotomy, we will lie in a Procrustian bed that is bound at times to be most uncomfortable. There must be degrees of being awake just as there are degrees of being asleep (i.e. the conventional sleep stages). Before finding our way out of this muddle, we will probably need to characterize a wider variety of states of consciousness than those few currently distinguished (e.g. 'dreaming,' 'sleeping,' 'waking,' and so on).
It may be helpful to consider lucidity from a cognitive developmental perspective. According to Piaget (1927), children pass through three stages of understanding of the concept "dream." In the first stage, they believe that dreams take place in the same external world as all other experiences. In the second stage, children treat dreams as if they were partially external and partially internal. This transitional stage gives way to the third stage in which children recognize the dream is entirely internal in nature, a purely mental experience.
These foregoing developmental stages refer to how children think about dreams when they are awake. While asleep and dreaming, children, and also adults, tend to remain at the first stage ¬ implicitly assuming that the dream events are external reality. Out-of-body experiences, with a contradictory mixture of material and mental (external and internal), may provide examples of the second stage (LaBerge, Levitan, Brylowski & Dement, 1988). In the fully lucid dream, the dreamer attains the third stage, realizing that the dream world is distinct from the physical world.
Foulkes (1982, 1985) has emphasised the idea that the growth of mind whether dreaming or awake shows parallel degrees of development: "...there are 'stages' of dream development which individual children reliably pass through one after the other, and that the precise age at which they reach a new stage is at least partially predictable from independent measures of their waking mental development." (1985, p. 137)
Lucid dreaming represents in this view what ought to be a normal ability in adults. If this is correct, why are lucid dreams so rare, especially in cases such as nightmares, where lucidity should be extremely helpful and rewarding? I think a possible answer can be seen by comparing lucid dreaming with another cognitive skill -- language. All normal adults speak and understand at least one language. But how many would do so if they were never taught? Unfortunately, in this culture, with few exceptions, we are not taught to dream.
LaBerge (1980b) has demonstrated that lucid dreaming is a learnable skill and there are a variety of techniques available for inducing lucid dreams (LaBerge, 1985; Price & Cohen, 1988). The Stanford group has experimented with methods for helping dreamers to realize that they are dreaming by means of external cues applied during REM sleep, which if incorporated into dreams, can remind dreamers that they are dreaming (LaBerge, 1980a). They have tested a variety of stimuli, including tape recordings of the phrase "This is a dream," (LaBerge, Owens, Nagel & Dement, 1981) conditioned tactile stimuli, (Rich, 1985) olfactory stimuli, (LaBerge, Brylowski & Levitan, 1986) and light (LaBerge, Levitan, Rich & Dement, 1988). The most promising results so far have been with light stimuli.
The psychophysiological studies reviewed above all support the following picture: During REM dreaming, the events we experience (or seem to) are the results of patterns of CNS activity that produce in turn effects on our ANS and bodies to some extent modified by the specific conditions of active sleep, but still homomorphic to the effects that would occur if we were actually to experience the corresponding events while awake.
This conclusion may need further qualification and explanation. Although the events we appear to perceive in dreams are illusory, our feelings in response to dream content are real. Indeed, most of the events we experience in dreams are real; when we experience feelings, say, anxiety or ecstasy, in dreams, we really do feel anxious or ecstatic at the time. When we think in dreams, we really do think (whether clearly or not is another matter). If we think in our dreams that Monday comes before Sunday, it is not the case, as some philosophers (e.g., Malcolm, 1959) assert, that we have only dreamed we thought; we may have thought incorrectly (to the usual way of thinking), but thought nonetheless.
If we were to vividly imagine a detailed sequence of movements, say, walking around the room, it is probable that motor areas of the brain would be activated in the same pattern as involved in actually walking. However, they would presumably be less activated than when walking. Otherwise, what would prevent us from actually walking when we imagined doing so?
In REM sleep there is a spinal paralysis which causes the muscles of locomotion and vocalization to fail to completely execute the action orders programmed by the brain. Thus, in REM, unlike the waking state, there is no impediment to the brain issuing sequences of motor commands at normal levels of activation, and this probably contributes to the experienced reality of dreamed action.
As for the afferent side of the equation, there is a great deal of evidence suggesting that imagery uses the same neural systems as perception in the corresponding sensory mode (see for example, Farah (1988) and Finke (1980). In this view, the essential difference between a perception and a corresponding image is how the identical neural system acquires sufficient activation to produce a conscious experience. In the case of perception, neural excitation (and the resultant experience) is generated by external input, driving activation of the particular schema to-be-perceived in a largely bottom-up process. In the case of imagining (likewise, hallucinating, or dreaming) the experienced image is generated internally by top-down processes activating the appropriate neural network (schema).
Imaginations and perceptions are normally distinguishable by the fact that images are usually much less vivid than perceptions. Normally, perceptions seem real and images seem -- imaginary. How real something appears depends mainly on its relative vividness and experienced vividness is probably a function of intensity of neural activation. Thus, we may conjecture that images usually involve a lesser degree of neural activation than the corresponding perceptions, and this results in a lesser degree of experiential reality for imagination. At least two factors contribute to this state of affairs: one is that while we are awake sensory input produces much higher levels of activation than imaginary input. Imagination interferes with perception in the same modality (Perky, 1910; Segal, 1971) and we may suppose the reverse is true as well. Another more speculative factor favoring perceptual processes over imagination in the waking state is the existence of a neural system to inhibit the activation (vividness) of memory images while perception is active. Evolutionary considerations make such a system likely; it would obviously be extremely maladaptive for an organism to mistake a current perceptual image of a predator for the memory of one (LaBerge, 1985). Mandell (1980) has implicated serotonergic neurons as part of a system that normally inhibits vivid images (hallucinations), but is itself inhibited in REM sleep, allowing dreamed perceptions (i.e., images) to appear as vividly real as perceptions. In REM, also, sensory input is actively suppressed preventing competition from perceptual processes.
Perhaps this explains in part why we are so inclined to mistake our dreams for reality: To the functional systems of neuronal activity that construct our experiential world (model), dreaming of perceiving or doing something is equivalent to actually perceiving or doing it.
Acknowledgements
I am grateful to the Institute for Human Development, the Mericos Foundation, and the Reality Foundation for financial support, and to Lynne Levitan for editorial assistance.
References
Anderson, J.R. (1983). The architecture of cognition. Cambridge: Harvard University Press.
Antrobus, J. S., Antrobus, J. S., & Fisher, C. (1965). Discrimination of dreaming and nondreaming sleep. Archives of General Psychiatry, 12, 395- 401.
Antrobus, J.S. (1986). Dreaming: Cortical activation and perceptual thresholds. Journal of Mind and Behavior, 7, 193-212.
Antrobus, J.S., Dement, W., & Fisher, C. (1964). Patterns of dreaming and dream recall: An EEG study. Journal of Abnormal and Social Psychology, 69, 244-252.
Arkin, A., Antrobus, J., & Ellman, S. (1978). (Eds.) The mind in sleep. Hillsdale, New Jersey: Lawrence Erlbaum Associates.
Aserinsky, E. (1971). Rapid eye movement density and pattern in the sleep of young adults. Psychophysiology, 8, 361-375.
Aserinsky, E., & Kleitman, N. (1953). Regularly occurring periods of eye motility and concomitant phenomena during sleep. Science, 118, 273-274.
Berger, R. (1977). Psyclosis: The circularity of experience. San Francisco: W. H. Freeman & Co.
Brylowski, A., Levitan, L., & LaBerge, S. (1989). H-reflex suppression and autonomic activation during lucid REM sleep: A case study. Sleep, 12, 374-378.
Dane, J. (1984). An empirical evaluation of two techniques for lucid dream induction. Unpublished doctoral dissertation, Georgia State University.
Dement, W., & Kleitman, N. (1957). Cyclic variations in EEG during sleep and their relation to eye movements, body motility, and dreaming. EEG and Clinical Neurophysiology, 9, 673-690.
Farah, M.J. Is visual imagery really visual? Overlooked evidence from neurophysiology. (1988). Psychological Review, 95, 307-317.
Fenwick, P., Schatzman, M., Worsley, A., Adams, J., Stone, S., & Baker, A. (1984). Lucid dreaming: Correspondence between dreamed and actual events in one subject during REM sleep. Biological Psychology, 18, 243-252.
Finke, R.A. (1980). Levels of equivalence in imagery and perception. Psychological Review, 87, 113-132.
Foulkes, D. (1974). [Review of Schwartz & Lefebvre (1973)]. Sleep Research, 3, 113.
Foulkes, D. (1980). Dreams and dream research. In W. Koella (Ed.), Sleep 1980. (pp. 246-257) Basel: Karger.
Foulkes, D. (1982). A cognitive-psychological model of dream production. Sleep, 5, 169-187.
Foulkes, D. (1985). Dreaming: A cognitive-psychological analysis. Hillsdale, New Jersey: Lawrence Erlbaum Associates.
Gackenbach, J., & LaBerge, S. (1988). Conscious mind, sleeping brain. New York: Plenum Press.
Garfield, P. (1979). Pathway to ecstasy. New York: Holt, Rhinehart, & Winston.
Green, C. (1968). Lucid dreams. London: Hamish Hamilton.
Hartmann, E. (1975). Dreams and other hallucinations: an approach to the underlying mechanism. In R. K. Siegal & L. J. West (Eds.), Hallucinations (pp. 71-79). New York: J. Wiley & Sons.
Hearne, K. M. T. (1978). Lucid dreams: An electrophysiological and psychological study. Unpublished doctoral dissertation, University of Liverpool.
LaBerge, S. (1980a). Lucid dreaming: An exploratory study of consciousness during sleep. (Doctoral dissertation, Stanford University, 1980). (University Microfilms International No. 80-24,691).
LaBerge, S. (1980b). Lucid dreaming as a learnable skill: A case study. Perceptual and Motor Skills, 51, 1039-1042.
LaBerge, S. (1980c). Induction of lucid dreams. Sleep Research, 9, 138.
LaBerge, S. (1985). Lucid dreaming. Los Angeles: J. P. Tarcher.
LaBerge, S. & Dement, W. C. (1982a). Voluntary control of respiration during REM sleep. Sleep Research, 11, 107.
LaBerge, S. and Dement, W. C. (1982b). Lateralization of alpha activity for dreamed singing and counting during REM sleep. Psychophysiology, 19, 331-332.
LaBerge, S., Brylowski, A. & Levitan, L. (1986). [unpublished data]
LaBerge, S., Greenleaf, W., & Kedzierski, B. (1983). Physiological responses to dreamed sexual activity during lucid REM sleep. Psychophysiology, 20, 454-455.
LaBerge, S., Levitan, L., & Dement, W. C. (1986). Lucid dreaming: Physiological correlates of consciousness during REM sleep. Journal of Mind and Behavior, 7, 251-258.
LaBerge, S., Levitan, L., Brylowski, A., & Dement, W. (1988). "Out-of-body" experiences occurring during REM sleep. Sleep Research, 17, 115.
LaBerge, S., Levitan, L., Rich, R., & Dement, W. (1988). Induction of lucid dreaming by light stimulation during REM sleep. Sleep Research, 17, 104.
LaBerge, S., Nagel, L., Dement, W. C., & Zarcone, V., Jr. (1981). Lucid dreaming verified by volitional communication during REM sleep. Perceptual and Motor Skills, 52, 727-732.
LaBerge, S., Nagel, L., Taylor, W., Dement, W. C., & Zarcone, V., Jr. (1981). Psychophysiological correlates of the initiation of lucid dreaming. Sleep Research, 10, 149.
LaBerge, S., Owens, J., Nagel, L., & Dement, W. (1981). "This is a dream": Induction of lucid dreams by verbal suggestion during REM sleep. Sleep Research, 10, 150.
Malcolm, N. (1959). Dreaming. London: Routledge.
Mandell, A.J. (1980). Toward a psychobiology of transcendence: God in the brain. In J.M. Davidson & R.J. Davidson (Eds.), The Psychobiology of Consciousness. New York: Plenum Press.
Ogilvie, R., Hunt, H., Kushniruk, A. & Newman, J. (1983). Lucid dreams and the arousal continuum. Sleep Research, 12, 182.
Ogilvie, R., Hunt, H., Sawicki, C., & McGowan, K. (1978). Searching for lucid dreams. Sleep Research, 7, 165.
Ogilvie, R., Hunt, H., Tyson, P.D., Lucescu, M. L. & Jeakins, D. B. (1982). Lucid dreaming and alpha activity: A preliminary report. Perceptual and Motor Skills, 55, 795-808.
Perky, C.W. (1910). An experimental study of imagination. American Journal of Psychology, 21, 422-452.
Piaget, J. (1926). The child's conception of the world. New York: Harcourt, Brace & Co. Pivik, R.T. (1986). Sleep: Physiology and psychophysiology. In M.G.H. Coles, E. Donchin, & S. Porges (Eds.). Psychophysiology: Systems, processes, and applications. Guilford Press: New York. 378-406.
Price, R. F. & Cohen, D. B. (1988). Lucid dream induction: An empirical evaluation. In J. Gackenbach & S. LaBerge (Eds.) Conscious mind, dreaming brain (pp. 105-154). New York: Plenum Press.
Rechtschaffen, A. & Kales, A. (Eds.). (1968). A manual of standardized terminology, techniques and scoring system for sleep stages of human subjects. Bethesda: HEW Neurological Information Network.
Rechtschaffen, A. (1978).The single-mindedness and isolation of dreams. Sleep, 1, 97-109.
Rich, R. (1985). The induction of lucid dreams by tactile stimulation during REM sleep. Unpublished honors thesis.
Roffwarg, H., Dement, W. C., Muzio, J., & Fisher, C. (1962). Dream imagery: Relationship to rapid eye movements of sleep. Archives of General Psychiatry, 7, 235-238.
Schwartz, B. A. & Lefebvre, A. (1973). Contacts veille/P.M.O. II. Les P.M.O. morcelees [Conjunction of waking and REM sleep. II. Fragmented REM periods.]. Revue d'Electroencephalographie et de Neurophysiologie Clinique, 3, 165-176.
Segal, S.J. (1971). Processing of the stimulus in imagery and perception. In S.J. Segal (Ed.) Imagery: Current cognitive approaches (pp. 73-100). New York: Academic Press.
Snyder, T. & Gackenbach, J. (1988). In J. Gackenbach & S. LaBerge (Eds.) Conscious mind, dreaming brain (pp. 221-259). New York: Plenum Press.
Stoyva, J. & Kamiya, J. (1968). Electrophysiological studies of dreaming as the prototype of a new strategy in the study of consciousness. Psychological Review, 75, 192-205.
Tart, C. (1988). From spontaneous event to lucidity: A review of attempts to consciously control nocturnal dreaming. In J. Gackenbach & S. LaBerge (Eds.), Conscious mind, dreaming brain (pp. 67-103). New York: Plenum Press.
Van Eeden, F. (1913). A study of dreams. Proceedings of the
Society for Psychical Research, 26, 431-461.